Bushra Quyoom and Latif Ahmad Peer, University of Kashmir, discuss their article: The Epigenetic Engine: Transgenerational plasticity as a driver of plant climate resilience and adaptation
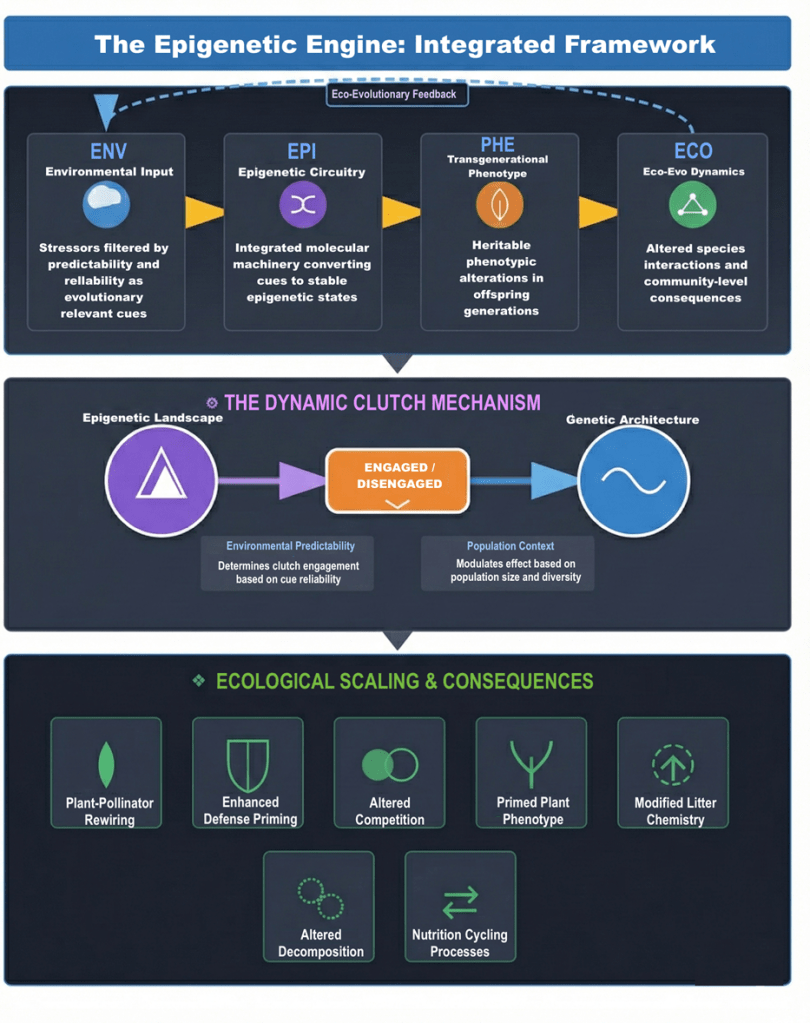
The traditional story of evolution is a slow one. We are taught that it can take thousands of years for helpful genetic mutations to appear and spread through a population. But today, the environment is changing at a breakneck pace. From heatwaves to unpredictable droughts, plants are facing an “adaptation debt”—a gap between how fast the world is changing and how well their DNA can keep up. We believe that plants have a secret weapon to bridge this gap: the Epigenetic Engine.
Beyond the DNA sequence
Most people think of inheritance as a fixed blueprint of A, T, C, and G. However, plants possess a sophisticated “memory” system that allows them to pass on experiences to their offspring without changing a single letter of their genetic code.
This system, known as Transgenerational Epigenetic Inheritance (TEI), works like a series of molecular switches. When a parent plant survives a drought or a pest attack, it doesn’t just adapt itself; it “reprogrammes” its seeds. Using a circuit of small RNAs, chemical tags on DNA (methylation), and protein scaffolds (histones), the plant sends a message to the next generation: “Be ready. The world is dry and dangerous.”
Why the “Engine” matters
We call this an “engine” because it is a regulated, environmentally responsive system that converts stress into survival strategies.
But what do we mean by “regulated”? This isn’t just a metaphor—it describes a system with built-in molecular controls. First, the engine has evolved “erasers”—enzymes like ROS1 and proteins like DDM1—that actively reset most epigenetic marks during seed development. This prevents plants from inheriting every random stress their parents have experienced. Only signals that are recurrent and ecologically relevant, like seasonal drought or repeated herbivore attacks, can survive this filtering process. Second, the response is locus-specific: methylation changes don’t occur randomly across the genome but target particular genes, such as the stress-responsive gene CNI1 in Arabidopsis. This precision ensures the system is adaptive rather than chaotic.
This regulation naturally raises a second question: are there trade-offs? Yes—and they are fundamental to understanding when and why the engine works. Three trade-offs are particularly important:
The mismatch cost: When parental and offspring environments differ, primed offspring can be less fit than unprimed ones. A plant primed for drought, if it grows in a wet year, may have invested resources in defence that could have gone into growth—a costly mistake.
Evolutionary inertia: The same epigenetic buffering that keeps a population alive through a crisis can, paradoxically, slow down genetic adaptation. By maintaining fitness without genetic change, epigenetic memory can relax selection pressure on the underlying DNA, potentially leaving populations dependent on a temporary fix.
Ecological trade-offs: Primed plants with tougher, chemical-laden leaves decompose more slowly, which can alter nutrient cycles in ways that may disadvantage their own offspring or competing species.
So, the epigenetic engine is not a magic bullet—it’s an adaptive strategy that works best when environmental challenges are predictable. Understanding these regulatory mechanisms and trade-offs is just as important as recognizing the engine’s power.
With this foundation, we propose that the engine serves two vital roles:
The Standalone Buffer: Rapid phenotypic tuning
Think of this as a “quick fix” that operates within one to two generations. When a parent plant experiences drought, it can transmit methylation marks that help offspring conserve water—without waiting for genetic mutations to arise. For example, rice plants exposed to multi-generation drought produced progeny with altered root architecture and better water-use efficiency even when those progeny never experienced drought themselves. Similarly, white clover subjected to herbivory produced offspring with denser trichomes and enhanced chemical defences. This buffer operates immediately because it uses existing epigenetic machinery, not rare genetic accidents.
The Dynamic Clutch: A temporal bridge to genetic evolution
The “clutch” metaphor comes from engineering—it engages or disengages power transfer. Here, epigenetic inheritance acts as a coupling between short-term survival and long-term evolution. When a population faces sudden climate stress, epigenetic buffering can maintain viability long enough for beneficial genetic mutations to arise and spread. A recent study on marine copepods exposed to warming and acidification found that epigenetic changes preserved genetic diversity during the initial crisis, allowing natural selection to later fix beneficial alleles once conditions had stabilized. In plants, heat-stressed Arabidopsis populations showed similar patterns: multigenerational exposure to high temperatures generates both epigenetic and genetic variation, with epigenetic changes appearing first and potentially paving the way for genetic accommodation. The clutch “engages” epigenetic memory during a crisis, then “disengages” to let genetics take over.
Scaling up: From one plant to an entire forest
In our review, we synthesized evidence from multiple studies to propose a new framework: the Transgenerational Ecosystem Legacy Hypothesis. This hypothesis connects molecular epigenetic changes to ecosystem-level processes.
Consider this scenario: dandelion plants (Taraxacum brevicorniculatum) exposed to intense competition transmit DNA methylation marks to their offspring. These offspring produce leaves with different chemical composition—specifically, higher fibre content and altered carbon-to-nitrogen ratios. When these leaves senesce and fall, they decompose 8–24% more slowly than leaves from unprimed plants.
Similarly, in red alder (Alnus rubra), herbivory on parent plants triggers transgenerational defence priming. The offspring produce leaves with 12.7% higher C:N ratios, which decompose 42% more slowly in adjacent streams, altering aquatic nutrient cycling.
Now scale this up: if an entire population becomes primed—through widespread drought, herbivore outbreaks, or competition—the cumulative effect on litter chemistry could shift decomposition dynamics across the landscape. Slower decomposition means nutrients are retained longer in recalcitrant organic matter, potentially favouring fungal-dominated decomposition pathways over bacterial ones. The “memories” of one generation literally reshape the chemical environment inherited by the next.
The future of resilience
The “adaptation debt”—the gap between the pace of environmental change and the speed of genetic evolution—is undoubtedly a heavy burden. But our review of the evidence suggests that plants are far more agile than classical evolutionary theory predicted. Through the epigenetic engine, they can draw on a reservoir of non-genetic memory: drought-primed rice remembers water scarcity, herbivore-attacked alder transmits chemical defences, and competition-exposed dandelions reshape the soil their offspring will grow in.
This doesn’t mean plants are invincible—the engine has limits and trade-offs, but it does mean that plants are not passive victims waiting for genetic mutations to rescue them. They are active participants in their own survival, transmitting the lessons of one generation to the next. Understanding this hidden language of inheritance—its power and its limits—may be key to helping plants, and us, navigate the challenges ahead. As we face a global climate crisis, this understanding offers a roadmap for “climate-smart” agriculture and conservation. If we can learn to prime this engine responsibly—by exposing seeds to mild stressors that match probable future conditions, or through emerging “epigenome editing” techniques—we might be able to help our crops and wild forests navigate a rapidly changing world. But we must proceed with eyes open, monitoring whether epigenetic solutions are buying time for genetic adaptation or inadvertently delaying it.


