Daniel Souto‐Vilarós tells us more about his recent Journal of Ecology paper on fig-wasp mutualisms. You can see Daniel present his work at Evolution 2018 – his poster is part of Session 1 and will be available to view up on Level 2 on August 19th and 20th (Poster number: P-0200, S-18).
Plant-pollinator interactions offer a unique arena for the study of evolution, speciation and character divergence. In the most specialized mutualisms, survival and reproduction of both parties depend almost exclusively on each other, suggesting strong selection pressures to ensure the ‘lock-and-key’ mechanisms which maintain this relationship.
In our recent paper in Journal of Ecology, we focus on the fig (Moraceae) and fig-wasp (Agaonidae) mutualism where pollinating wasps also lay eggs within the fig, which serves as a nursery for the next generation of pollinators.
We studied a three sister species complex (Ficus itoana, F. sp. and F.microdictya) and both subspecies of Ficus trichocerasa, along the Mt. Wilhelm elevational transect in Papua New Guinea, the centre of Ficus diversity (fig. 1). All of our focal species are dioecious except for Ficus microdictya which is one of the few known cases of reversal from dioecy to monecy in Ficus.

Figure 1. Geographical location of collection sites along the Mount Wilhelm elevational gradient in Madang province, Papua New Guinea. Contour lines very 100 m. Inset: Focal Ficus species used in this study and their corresponding fig wasps: a: Ficus itoana; b: Ficus sp.; c: Ficus microdictya; d: Ficus trichocerasa.
With over 750 described species, the maintenance of species specificity between figs and fig-wasps is believed to depend primarily on unique volatile signals which attract fig-wasps to their host fig species. We compared these volatile organic compounds (VOCs) from figs of our focal species and using high-throughput sequencing, we placed these species in a phylogenetic context. Using Y-tube assays we tested fig-wasp responses to volatiles emitted by its host species, as well as volatiles from closely related figs in order to detect a scent preference by these wasps.
We found that closely related fig species have significantly different scent profiles, and indeed even subspecies have different VOC compositions from each other. However, scent alone appeared not to be enough for complete segregation of pollinating wasps. For two of the three wasp species tested, wasps consistently preferred the emissions from its host fig instead of air and air instead of a non-host species, as expected. However, fig-wasps which emerged from F. sp., an as of yet undescribed species restricted to the mid elevations (~ 1700m above sea level), were attracted to both its host, and its host’s sister species, Ficus microdictya, which is restricted to altitudes above 2,200m.
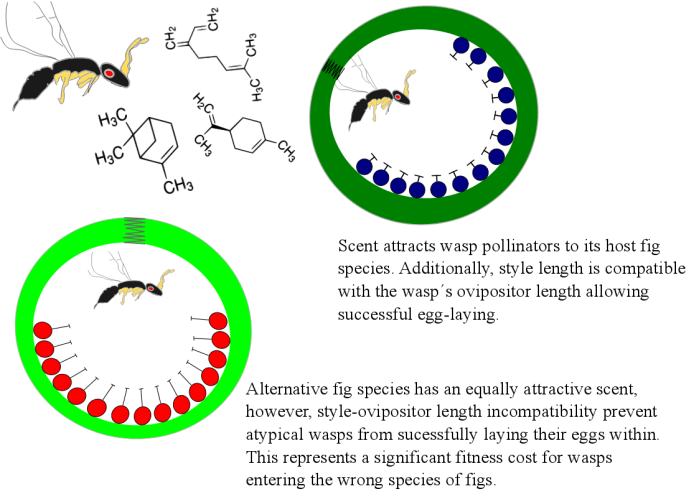
Figure 2. Summary of the pollination barriers which help maintain pollinator isolation in the fig and fig-wasp mutualism in case volatile signals remain equally attractive.
This result suggests that in the event of a fig-wasp from F. sp. would drift uphill, it could be attracted to the ‘wrong’ fig species. In this case, secondary prepollination barriers act in order to maintain reproductive isolation between both fig species, and thus, the pollinating wasps. Fig size at receptivity is known to be correlated with wasp head morphology; this is because fig-wasps must crawl through a tiny cavity at the apex of the fig (called ostiole) before reaching the flowers enclosed within. Wasps from F. sp. have significantly larger heads than those from F. microdictya suggesting that head size could prevent these wasps from entering F. microdictya figs. Nevertheless, if the wasp is determined to enter the fig, a final barrier may present itself.
In dioecious figs, male trees have exclusively short-styled flowers which match the size of the wasp’s ovipositor. The wasp will crawl through the ostiole and while pollinating all available flowers, she simultaneously lays eggs within the ovules; In female figs, the wasp is tricked to enter the fig where she finds only long-styled flowers and though possible to pollinate, the wasp in unable to oviposit in the flowers and so the fig produces only seeds.
In the case of monoecious species, there is a mix of long- and short-styled flowers usually compatible with the ovipositor length of its pollinating fig-wasps and so a portion of flowers will produce seeds while the others will produce the next generation of wasps. Ceratosolen “kaironkensis”, pollinator of F. microdictya has an ovipositor nearly twice as long as that of F. sp.’s pollinator indicating that if these wasps enter F. microdictya’s figs, they will be unable to oviposit in the flowers and die within the fig, representing a considerable fitness cost to the wasp.
In conclusion, our study reveals the multiple pollination barriers at play even in highly specific, obligate mutualisms. Although scent appears to be one of the most important mechanisms for attracting pollinators, character divergence in other traits (such as breeding system in figs, or wasp morphology; see fig. 2) further strengthen pollinator isolation and thus maintaining species specificity in this system.
Daniel Souto‐Vilarós, University of South Bohemia České Budějovice, Czech Republic
Read the full paper online: Pollination along an elevational gradient mediated both by floral scent and pollinator compatibility in the fig and fig‐wasp mutualism, and go and see Daniel’s poster at Evolution!


